Bohr Model and Its Applications
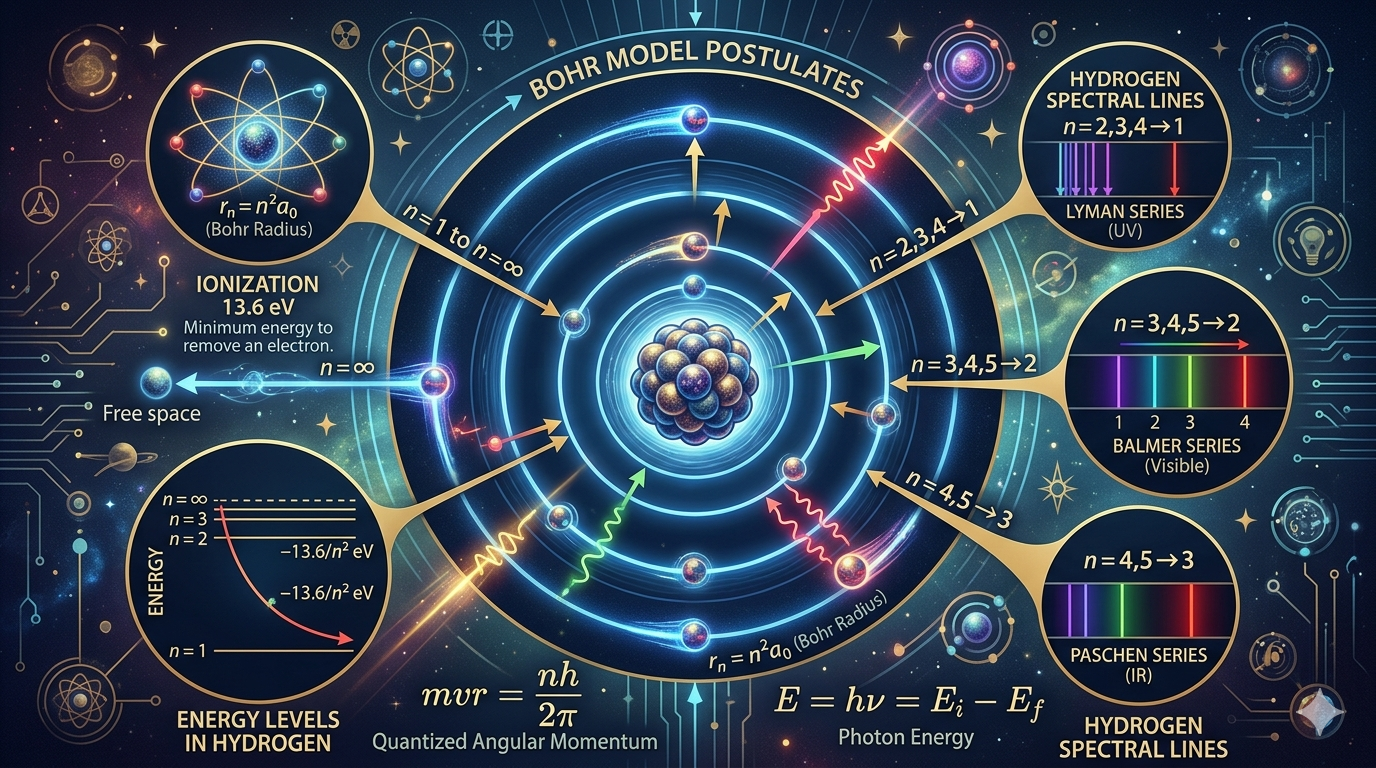
Image generated by Google AI
What Is Bohr Model?
Have you ever noticed the glow of a neon sign or the sharp colors in a hydrogen discharge tube in a lab? Those beautiful colors are not random, they are fingerprints of atoms. Each color corresponds to a very specific energy transition happening deep inside the atom. Now imagine this: an electron is not just wandering aimlessly but is restricted to very special “allowed” paths. This is exactly where the Bohr Model steps in.
The Bohr Model describes the hydrogen atom by combining classical mechanics with quantum ideas. It explains the discrete spectral lines observed in atomic emission.
Key Concept: Electrons revolve in specific orbits without radiating energy and transition between these levels by absorbing or emitting photons.
Deep Insight: Classically, any charged particle moving in a circular path should continuously radiate energy and spiral into the nucleus. But atoms are stable! Bohr’s bold assumption, that only certain orbits are allowed, quietly introduces quantization. In modern quantum mechanics, this emerges naturally from wave behavior of electrons, where only standing wave patterns are allowed around the nucleus.
Postulates of Bohr's Model
- Electrons orbit the nucleus in stable, quantized paths.
Think of these orbits like allowed “resonant tracks,” similar to how only certain musical notes sound stable on a guitar string.
- Angular momentum is quantized:
\[ m v r = \frac{n h}{2\pi} \]
Subtle Point: This is not just a rule, it hints that angular momentum comes in discrete packets. Later, de Broglie explained this as a condition for standing waves: \( 2\pi r = n\lambda \).
- Radiation is emitted/absorbed only during transitions:
\[ E = h \nu = E_i - E_f \]
Physical Meaning: The atom does not “gradually” lose energy. It jumps abruptly between levels, this is the origin of quantum jumps.
Energy Levels in Hydrogen Atom
\[ E_n = -\frac{13.6}{n^2} \, \text{eV} \]
Where \( n \) is the principal quantum number. Negative energy indicates the electron is bound.
Deep Physical Meaning: Zero energy corresponds to a free electron infinitely far away. So negative energy means the electron is trapped in the “potential well” of the nucleus, like a marble sitting inside a bowl.
Radius of orbit: \( r_n = n^2 a_0 \), where \( a_0 = 0.529 \, \text{Å} \)
Subtle Observation: As \( n \) increases, radius grows as \( n^2 \), but energy changes as \( 1/n^2 \). This asymmetry reflects how potential energy dominates at small distances while kinetic energy becomes weaker farther out.
Spectral Lines
Electron transitions lead to emission or absorption of light:
\[ \frac{1}{\lambda} = R_H \left( \frac{1}{n_1^2} - \frac{1}{n_2^2} \right) \]
Imagine this: Each jump between orbits is like a step on a staircase where the height difference determines the color of light emitted.
Hydrogen Series:
- Lyman (\( n_1 = 1 \)): UV
- Balmer (\( n_1 = 2 \)): Visible
- Paschen (\( n_1 = 3 \)): IR
Deep Insight: The discreteness of spectral lines is direct evidence that energy levels are quantized. If energy were continuous, we would observe a continuous spectrum instead of sharp lines.
Key Points
- Energy is quantized; levels are negative.
- \( E_n \propto 1/n^2 \), \( r_n \propto n^2 \)
- Only hydrogen or hydrogen-like ions fit the model.
- Photon frequency depends on energy gap.
Subtle Connection: The inverse-square dependence of energy comes directly from Coulomb’s law. This is why Bohr’s model works perfectly for hydrogen-like systems but fails when electron-electron interactions complicate the potential.
Example
Find the energy needed to ionize a hydrogen atom from its ground state.
\[ E_1 = -13.6 \, \text{eV}, \quad E_\infty = 0 \Rightarrow \text{Ionization energy} = 0 - (-13.6) = 13.6 \, \text{eV} \]
Answer: 13.6 eV
Physical Interpretation: This is the exact energy required to completely “liberate” the electron from the atom, taking it from a bound state to a free state.
Concept Questions
- Why are energy levels negative?
The electron is bound to the nucleus.
Insight: Negative energy reflects confinement in a potential well. - What happens when an electron absorbs energy?
It jumps to a higher orbit (excitation).
Subtlety: The jump is instantaneous—no intermediate states exist. - Can Bohr’s model explain multi-electron atoms?
No, only single-electron systems.
Reason: Electron-electron repulsion breaks the simple quantization. - How does radius change with \( n \)?
Increases as \( n^2 \). - What does Bohr's model explain?
Atomic spectra of hydrogen.
Super Tips
- Energy formula: \( E_n = -13.6/n^2 \, \text{eV} \). Lower \( n \) means more tightly bound electron.
- Radius formula: \( r_n = n^2 a_0 \). Orbit size increases rapidly with \( n \).
- Photon emission: Occurs when electron falls from higher to lower level:
\[ h \nu = E_i - E_f \]
Insight: Higher energy gap → higher frequency → shorter wavelength.
- Ionization energy: From ground state: 13.6 eV.
- Transition shortcuts:
Maximum energy: \( n = 2 \to 1 \)
Visible light: Balmer series
UV: Lyman series - Rydberg formula tip:
\[ \frac{1}{\lambda} = R_H \left( \frac{1}{n_1^2} - \frac{1}{n_2^2} \right) \]
- Electron speed: Decreases with increasing \( n \): \( v_n \propto 1/n \)
Subtle Point: As the electron moves farther, the Coulomb attraction weakens, reducing its speed.
- Model limitation: Works only for single-electron atoms like H, He⁺, Li²⁺.
Previous Year Question (PYQ)
Q1. A particle of mass \( m \) is moving around the origin with a constant force \( F \) pulling it towards the origin. If Bohr model is used to describe its motion, the radius of the \( n^{th} \) orbit and speed \( v \) depend on \( n \) as:
(NEET 2025)
- \( r \propto n^{2/3},\ v \propto n^{1/3} \)
- \( r \propto n^{4/3},\ v \propto n^{-1/3} \)
- \( r \propto n^{1/3},\ v \propto n^{1/3} \)
- \( r \propto n^{1/3},\ v \propto n^{2/3} \)
Solution:
Step 1: Use central force model
Given: A constant central force \( F \) pulls the particle towards origin.
Centripetal force: \( \frac{mv^2}{r} = F \Rightarrow v^2 \propto r \)
\[ \Rightarrow v \propto \sqrt{r} \tag{1} \]
Step 2: Apply Bohr's quantization condition
Angular momentum quantization:
\[ m v r = n \hbar \Rightarrow v \propto \frac{n}{r} \tag{2} \]
Substitute (1) into (2):
\[ \sqrt{r} \propto \frac{n}{r} \Rightarrow r^{3/2} \propto n \Rightarrow r \propto n^{2/3} \]
Now from (1): \( v \propto \sqrt{r} \Rightarrow v \propto n^{1/3} \)
Answer: \( r \propto n^{2/3},\ v \propto n^{1/3} \)
Deep Insight: Notice how the nature of force (constant vs inverse-square) completely changes the scaling laws. This shows that Bohr quantization is flexible—but the physics of the force determines the structure of allowed orbits.