Viscosity and Buoyancy
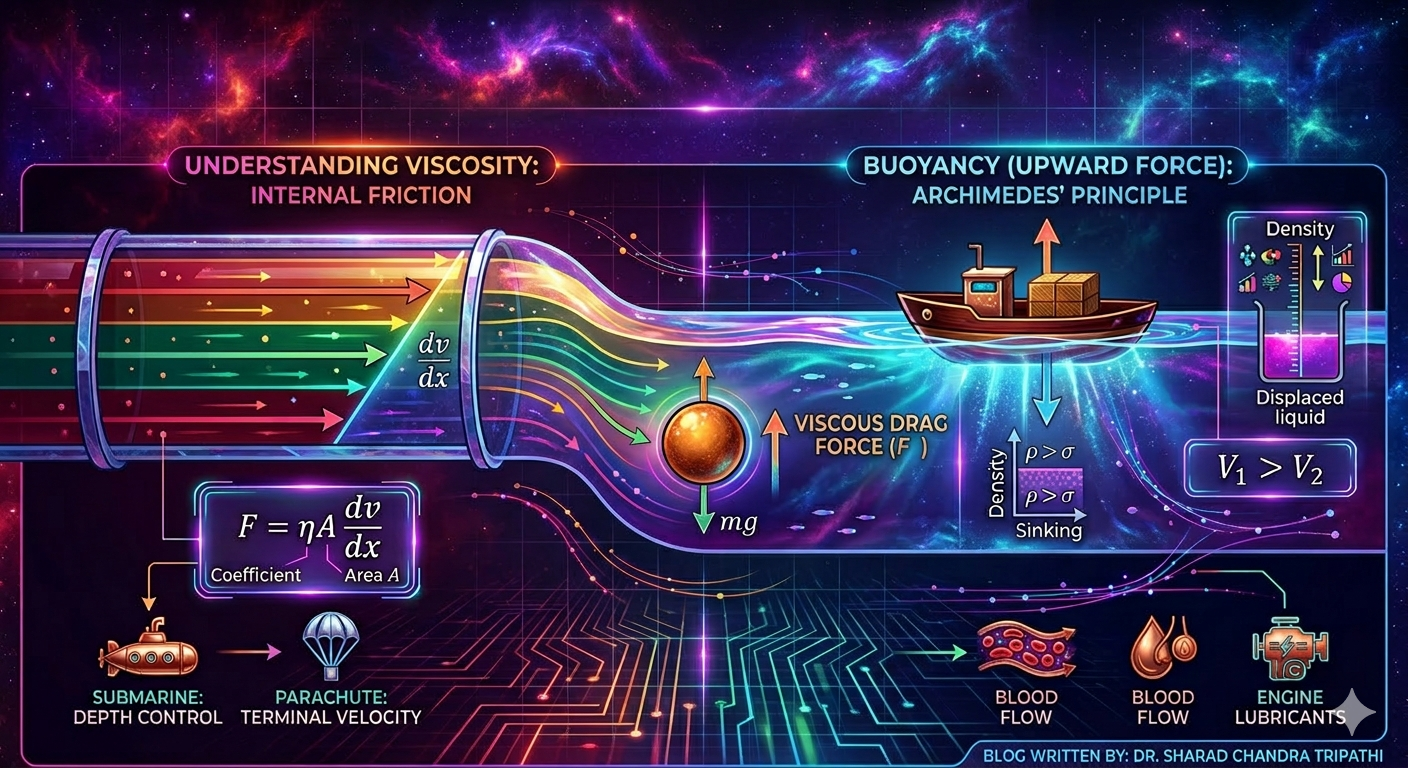
Image generated by Google AI
Have you ever wondered why honey flows more slowly than water? Or why a ball dropped in oil sinks more slowly than in air? If you have ever watched syrup being poured and felt like time itself slowed down for a moment, you have already observed physics in action. The answer lies in a property called viscosity. And when you think of floating objects or things submerged in water, that is where buoyancy comes in.
These two concepts, viscosity and buoyancy, are crucial in fluid mechanics and have real-life applications, from designing submarines to understanding blood flow in veins. In fact, every time your heart pumps blood, it quietly negotiates with viscosity, and every ship floating in the ocean is in constant conversation with buoyancy. Let us dive in.
What is Viscosity?
Viscosity is a measure of a fluid’s resistance to flow. Simply put, it is how “thick” or “sticky” a fluid is. Honey, oil, and glycerin have high viscosity. Water and air have low viscosity.
If a fluid flows easily, it has low viscosity. If it flows slowly, it has high viscosity. But here is something interesting, this “thickness” is not about how heavy a fluid is, but how strongly its molecules interact with each other while moving.
Why does this happen?
Because in fluids, different layers slide over each other when the fluid flows. Viscosity is the internal friction between these layers. On a microscopic level, molecules are constantly colliding, pulling, and resisting motion; almost like tiny dancers trying to stay in sync but slightly dragging each other back.
Viscous Force
When a fluid flows over a surface or within itself, a force opposes this motion. This is called the viscous force. It is nature’s way of saying, “motion is allowed, but not without resistance.”
Imagine two parallel layers of a fluid, one moving faster than the other. The layer below resists the motion of the one above, much like friction. If you could zoom in, you would see a smooth gradient of speeds, no sudden jumps, just a continuous change, which is a beautiful example of how nature avoids abruptness.
Mathematically, viscous force \( F \) is given by:
\[ F = \eta A \frac{dv}{dx} \]
Where:
- \( \eta \) is the coefficient of viscosity
- \( A \) is the area of the layer
- \( \frac{dv}{dx} \) is the velocity gradient (rate of change of velocity with distance)
That small term \( \frac{dv}{dx} \) carries a deep idea, it tells us that fluids care not just about speed, but about how speed changes from one layer to another. Physics often hides big ideas in small symbols.
Unit of viscosity: \( \text{Pa} \cdot \text{s} \) or \( \text{N} \cdot \text{s/m}^2 \)
CGS unit: Poise, where \( 1 \, \text{Pa} \cdot \text{s} = 10 \, \text{Poise} \)
Types of Fluids
- Ideal Fluid: No viscosity (purely theoretical, like a perfect world where nothing resists motion)
- Real Fluid: Has viscosity (everything around us)
- Newtonian Fluid: Obeys Newton’s law of viscosity (e.g., water, air)
- Non-Newtonian Fluid: Does not obey Newton’s law (e.g., ketchup, blood)
A fun twist: non-Newtonian fluids can behave strangely. Ketchup refuses to flow until you shake it, and then suddenly it does. Blood changes its viscosity depending on how fast it flows. These are reminders that nature does not always follow simple rules, it adapts.
Stoke's Law: Drag on a Sphere
Ever seen a ball falling in oil? It moves slowly because of viscous drag. It is almost as if the fluid is gently holding it back, not letting it rush.
When a sphere moves through a viscous fluid, it experiences a resisting force given by:
\[ F = 6 \pi \eta r v \]
- \( \eta \) = viscosity of the fluid
- \( r \) = radius of the sphere
- \( v \) = velocity of the sphere
Notice how the force increases with velocity. The faster you try to move, the stronger the fluid pushes back. This is very different from solid friction, which often stays constant. Fluids respond dynamically.
This law is valid for small spherical objects moving slowly (laminar flow). In this regime, the flow is smooth and predictable, almost like layers of silk sliding over each other.
Terminal Velocity
As an object falls through a fluid, it speeds up until the net force becomes zero. At this point, it moves with constant speed. This is called terminal velocity.
At first, gravity dominates. Then resistance builds up. Eventually, a perfect balance is reached, a quiet equilibrium where forces cancel out, and motion continues without acceleration. Physics often settles into balance.
For a sphere of radius \( r \), terminal velocity \( v_t \) is:
\[ v_t = \frac{2r^2(\rho - \sigma)g}{9\eta} \]
- \( \rho \) = density of the sphere
- \( \sigma \) = density of fluid
- \( g \) = acceleration due to gravity
- \( \eta \) = viscosity of fluid
So, if the object is denser than the fluid, it sinks. If lighter, it floats. And if the densities are close, motion becomes slow and graceful, almost suspended in time.
Buoyancy: The Upward Force
Have you ever felt lighter in a swimming pool? That is because of buoyancy, the upward force exerted by a fluid on any object placed in it. It is one of those rare moments where physics makes you feel like gravity has loosened its grip on you.
This concept was first explained by Archimedes over 2000 years ago. Legend says he discovered it while taking a bath, proof that even relaxed moments can lead to deep insights.
Archimedes’ Principle:
When a body is immersed in a fluid, it experiences an upward force equal to the weight of the fluid displaced.
\[ F_B = \text{Weight of displaced fluid} = \sigma V g \]
- \( \sigma \) = density of fluid
- \( V \) = volume of fluid displaced
- \( g \) = acceleration due to gravity
This idea connects force with displacement, when you push fluid out of the way, it pushes back. It is a subtle symmetry in nature.
Apparent Weight
\[ \text{Apparent weight} = \text{Actual weight} - \text{Buoyant force} \]
That is why lifting a heavy object underwater feels easier. You are not weaker outside water, the fluid is simply helping you.
Conditions for Floating and Sinking
- If \( \rho < \sigma \): The object floats.
- If \( \rho > \sigma \): The object sinks.
- If \( \rho = \sigma \): The object remains suspended.
This is also why massive ships float, they are designed so that their average density is less than water. It is not just material, but structure that matters.
Viscosity and Temperature
- For liquids: Viscosity decreases with temperature.
- For gases: Viscosity increases with temperature.
That is why hot honey flows faster than cold honey. When heated, molecules gain energy and overcome internal attractions more easily. In gases, however, faster molecules collide more often, increasing resistance, an interesting contrast that shows how the same concept behaves differently in different states.
Reynolds Number: Laminar vs Turbulent Flow
We use the Reynolds number \( Re \):
\[ Re = \frac{\rho v D}{\eta} \]
- \( \rho \) = density of fluid
- \( v \) = velocity
- \( D \) = characteristic length
- \( \eta \) = viscosity
Critical values:
- \( Re < 2000 \): Laminar flow (smooth)
- \( Re > 3000 \): Turbulent flow (chaotic)
- In between: Transitional
Reynolds number is like a “personality test” for fluid flow. Low values mean calm, orderly motion. High values mean chaos, eddies, and swirls. Somewhere in between, the fluid decides what it wants to be.
Applications in Real Life
- Designing vehicles to reduce drag (cars, airplanes, even bicycles)
- Blood flow in arteries (blood is a non-Newtonian fluid)
- Oil and lubricant selection in engines
- Designing parachutes and air-drops
Even nature uses these principles, fish have streamlined bodies to minimize drag, and birds instinctively adjust flow patterns while flying.
Shortcut Concepts
- Viscosity is internal friction in fluids.
- Viscous force: \( F = \eta A \frac{dv}{dx} \)
- Stoke’s Law: \( F = 6\pi \eta r v \)
- Terminal velocity: \( v_t = \frac{2r^2(\rho - \sigma)g}{9\eta} \)
- Buoyant force: \( F_B = \sigma V g \)
- Apparent weight = Actual weight - Buoyancy
- Reynolds number decides flow type: Laminar or Turbulent
If you remember just these, you already understand a large part of fluid mechanics.
Quick Review Questions
- What happens to viscosity of a liquid when it is heated?
It decreases. - A ball falls slowly in glycerin. What force is acting against gravity?
Viscous drag force and buoyant force. - Why do objects feel lighter in water?
Because of the upward buoyant force. - Which has higher viscosity: water or honey?
Honey. - What does Reynolds number signify?
It tells us whether fluid flow is laminar or turbulent.
Previous Year Question (PYQ)
Q.1
The diameter of an air bubble which was initially \(2 \, \text{mm}\), rises steadily through a solution of density \(1750 \, \text{kg/m}^3\) at the rate of \(0.35 \, \text{cm/s}\). The coefficient of viscosity of the solution is ........ poise (to nearest integer).
(The density of air is negligible.) [JEE, 2022]
Notice something subtle here, the bubble rises instead of falling. That is buoyancy winning over weight, while viscosity quietly limits how fast it can rise. This is a perfect combination of both concepts.
Solution:
Given values
- Diameter \(D = 2 \, \text{mm}\) ⇒ Radius \(r = 1 \, \text{mm} = 1 \times 10^{-3} \, \text{m}\)
- Terminal velocity \(v = 0.35 \, \text{cm/s} = 0.0035 \, \text{m/s}\)
- Density of solution \( \rho = 1750 \, \text{kg/m}^3 \)
- Density of air is negligible: \( \sigma \approx 0 \)
- Acceleration due to gravity \( g = 9.8 \, \text{m/s}^2 \)
- \( \pi = \frac{22}{7} \)
Use Stoke’s law and buoyant force equilibrium
\[ F_{\text{viscous}} = F_{\text{buoyant}} = V \rho g \]
\[ F = 6\pi \eta r v \quad \text{and} \quad V = \frac{4}{3} \pi r^3 \]
\[ 6\pi \eta r v = \frac{4}{3} \pi r^3 \rho g \]
\[ \eta = \frac{2}{9} r^2 \rho g / v \]
This step is where everything comes together, forces balancing, geometry entering through volume, and motion settling into a steady state.
Plug in the values
\[ \eta = \frac{2}{9} \cdot (1 \times 10^{-3})^2 \cdot 1750 \cdot 9.8 / (0.0035) \]
\[ = \frac{2 \cdot 10^{-6} \cdot 1750 \cdot 9.8}{9 \cdot 0.0035} = \frac{34.3 \cdot 10^{-3}}{0.0315} \approx 1.089 \, \text{Ns/m}^2 \]
Convert to poise:
\[ 1 \, \text{Ns/m}^2 = 10 \, \text{poise} \Rightarrow \eta \approx 10.89 \, \text{poise} \]
Answer (nearest integer): \( \boxed{11} \, \text{poise} \)